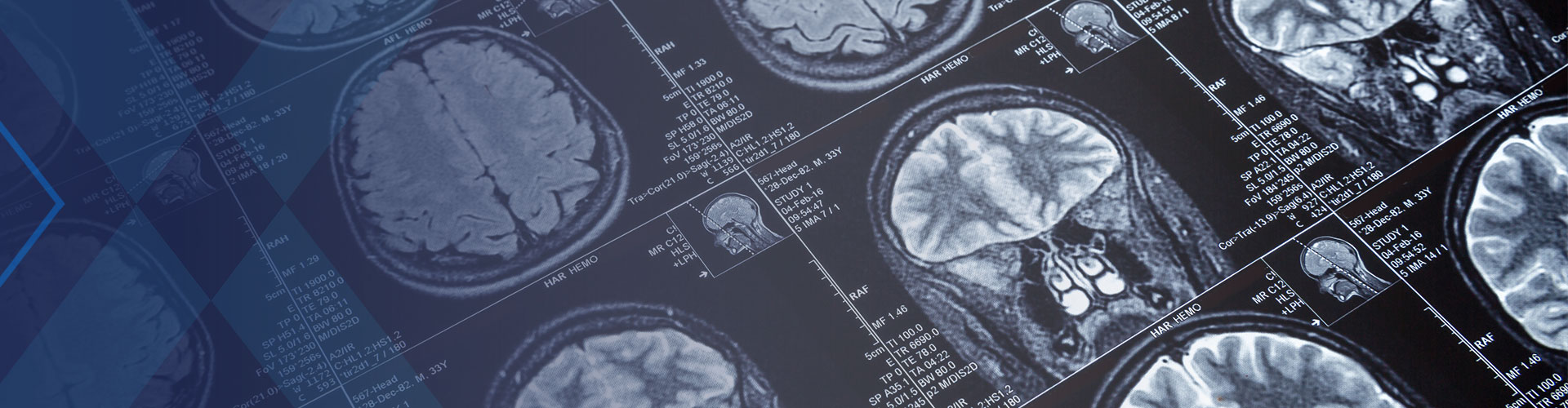
EP102 For MIGRAINE
Epalex is developing EP102 for the acute treatment of migraine headache pain and migraine-related symptoms. EP102 is an oral dosage form that can achieve circulating levels of a drug substance that has been approved by the FDA for a different indication. Until now, this drug could be administered only intravenously in an emergency room or inpatient setting.
The mechanism of action of EP102 differs from that of other products currently marketed for the acute treatment of migraine. EP102 is thought to impact migraine by modulating the inhibitory action of the neurotransmitter gamma aminobutyric acid (GABAA) on GABAA receptors and may be involved in the inhibition of the neurologic process of cortical spreading depression, a possible cause of migraine and migraine aura.
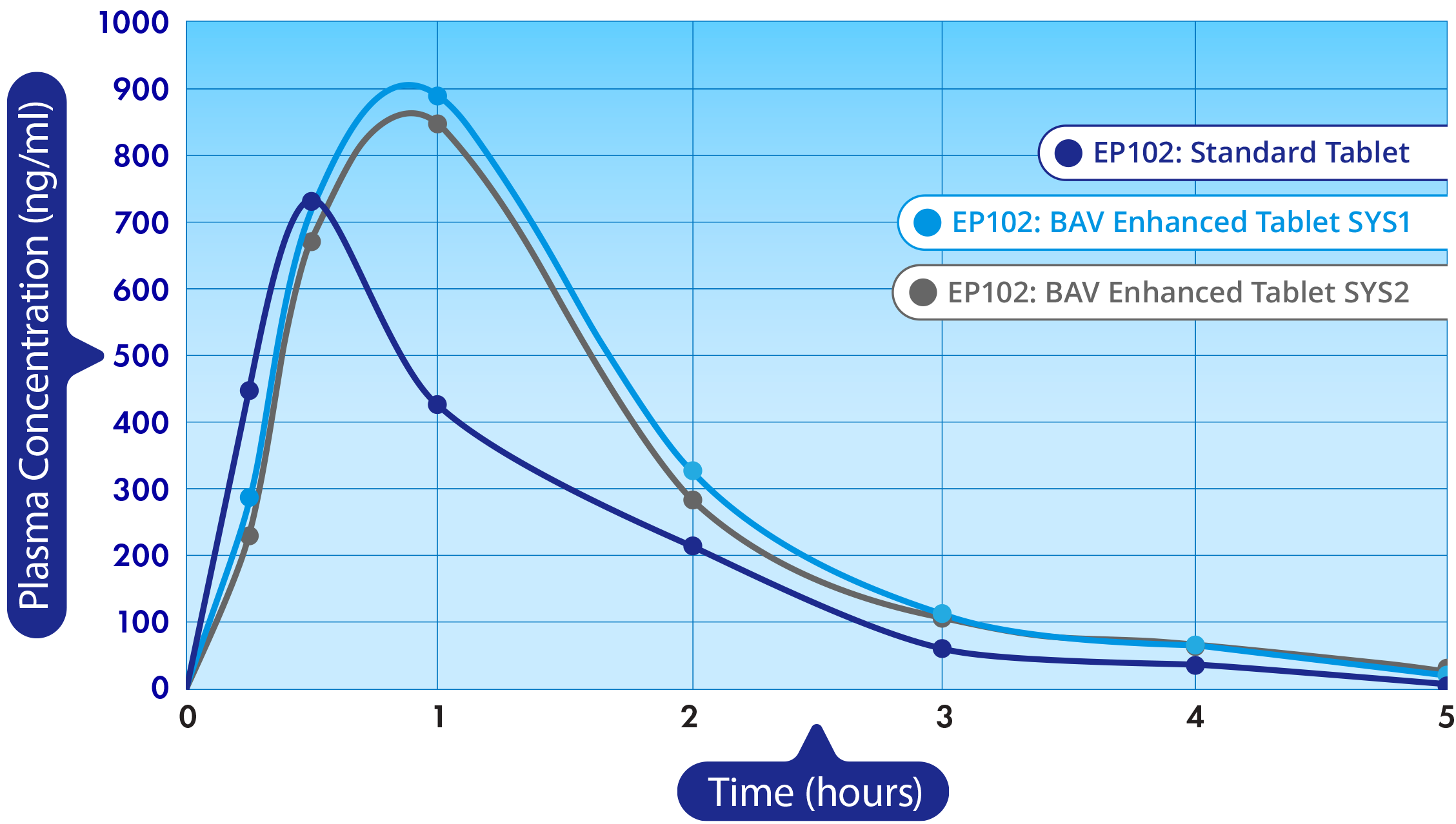
Epalex recently completed a Phase 1a, randomized, placebo-controlled, double-blind ascending-dose study of EP102 in 99 healthy male and female adults. This study established a well-characterized pharmacokinetic and safety profile, exhibiting good safety-tolerability of EP102.
A proof-of-concept study is currently underway using EP102 in adult women and men experiencing moderate to severe migraine.